Selected Publications

Persistent complement dysregulation with signs of thromboinflammation in active Long Covid
Cervia-Hasler C, Brüningk SC, Hoch T, Fan B, Muzio G, Thompson RC, Ceglarek L, Meledin R, Westermann P, Emmenegger M, Taeschler P, Zurbuchen Y, Pons M, Menges D, Ballouz T, Cervia-Hasler S, Adamo S, Merad M, Charney AW, Puhan M, Brodin P, Nilsson J, Aguzzi A, Raeber ME, Messner CB, Beckmann ND, Borgwardt K, Boyman O
Science (2024)
DOI: 10.1126/science.adg7942
Abstract
Acute infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes various clinical phenotypes, ranging from asymptomatic to life-threatening COVID-19. About 5% of all infected individuals do not recover from acute disease but develop long-term complications, called Long Covid. Current hypotheses on factors contributing to Long Covid include tissue damage, viral reservoirs, autoimmunity, and persistent inflammation. There are currently no diagnostic tests or therapeutic solutions for affected patients.Our data suggest that active Long Covid is accompanied by a blood protein signature marked by increased complement activation and thromboinflammation, including activated platelets and markers of red blood cell lysis. Tissue injury may also be complement-mediated and, in turn, activate the complement system. Moreover, complement activation may be driven by antigen–antibody complexes, involving autoantibodies and antibodies against herpesviruses, as well as cross-talk with a dysregulated coagulation system. In addition to offering a basis for new diagnostic solutions, our work provides support for clinical research on complement modulators for patients suffering from Long Covid.

Human memory B cells show plasticity and adopt multiple fates upon recall response to SARS-CoV-2
Zurbuchen Y, Michler J, Taeschler P, Adamo S, Cervia C, Raeber ME, Acar IE, Nilsson J, Warnatz K, Soyka MB, Moor AE, Boyman O
Nature Immunology (2023)
DOI: 10.1038/s41590-023-01497-y
Abstract
The B cell response to different pathogens uses tailored effector mechanisms and results in functionally specialized memory B (Bm) cell subsets, including CD21+ resting, CD21–CD27+ activated and CD21–CD27– Bm cells. The interrelatedness between these Bm cell subsets remains unknown. Here we showed that single severe acute respiratory syndrome coronavirus 2-specific Bm cell clones showed plasticity upon antigen rechallenge in previously exposed individuals.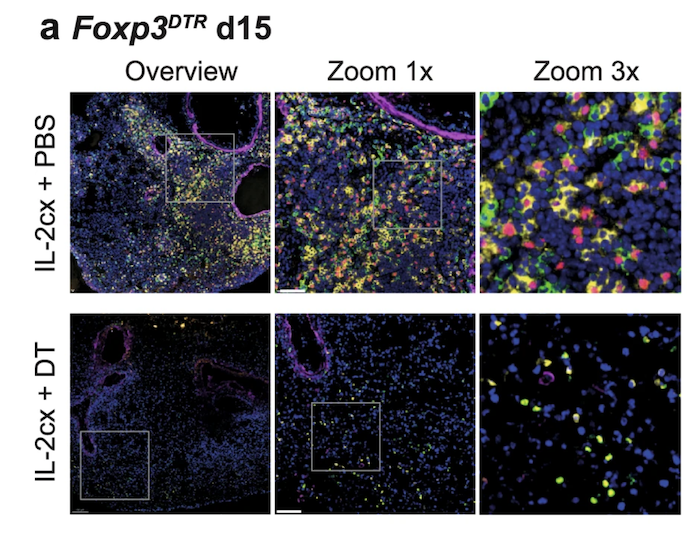
Biased IL-2 signals induce Foxp3-rich pulmonary lymphoid structures and facilitate long-term lung allograft acceptance in mice
Yamada Y, Nguyen T, Impellizzieri D, Mineura K, Shibuya R, Gomariz A, Haberecker M, Nilsson J, Nombela-Arrieta C, Jungraithmayr W, Boyman O
Nature Communications (2023)
DOI: 10.1038/s41467-023-36924-z
Abstract
Insight into the pathomechanism of atopic diseases demonstrated a pivotal role of the cytokines interleukin-4 (IL-4) and IL-13, which has spurred the development of tailored therapeutics targeting their common IL-4 receptor (IL-4R). However, several aspects of the IL-4R system remain ill-defined in humans.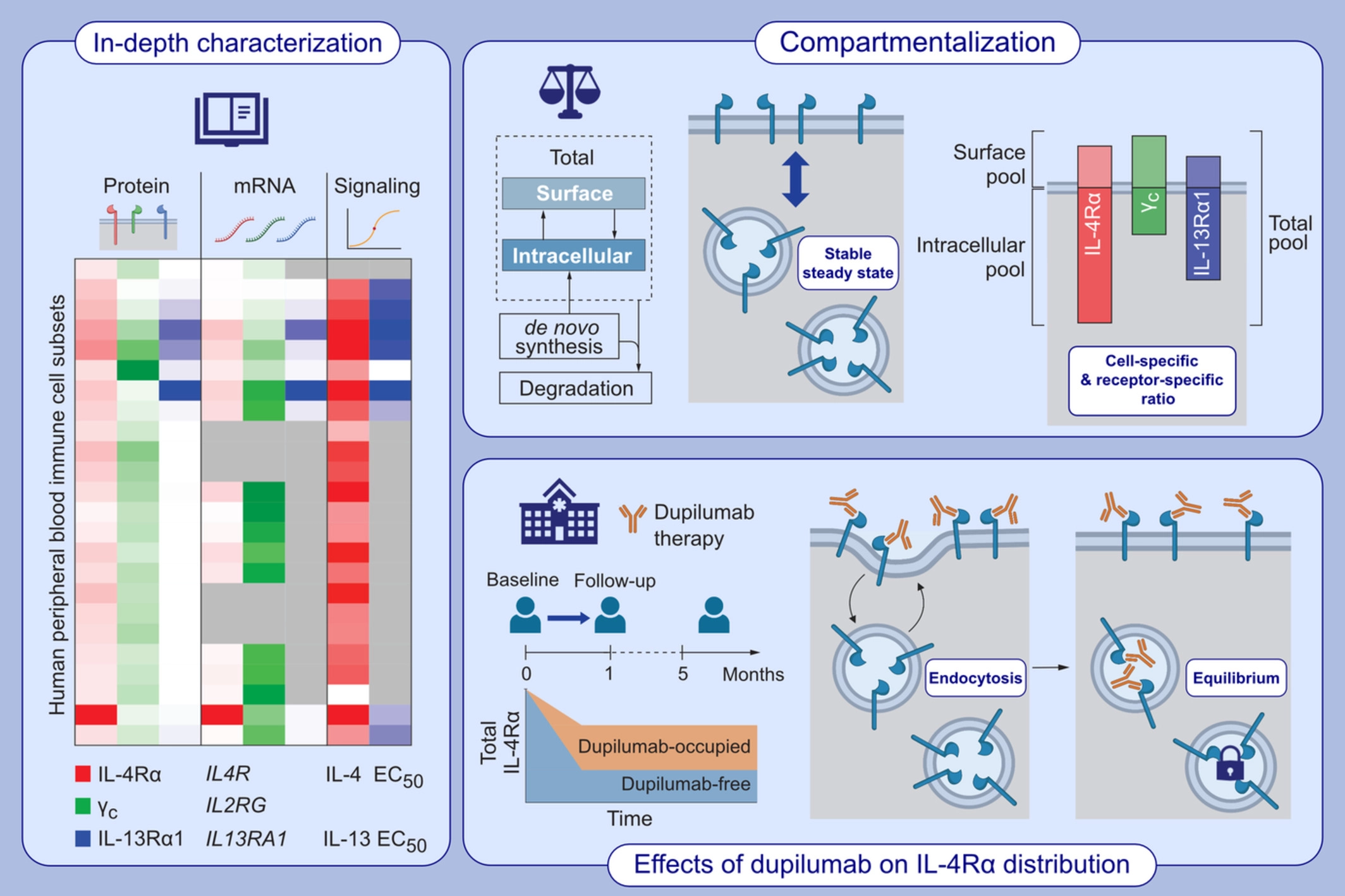
Comprehensive analysis of human IL-4 receptor subunits shows compartmentalization in steady state and dupilumab treatment
Heeb L, Boyman O
Allergy (2022)
DOI: 10.1111/all.15576
Abstract
Insight into the pathomechanism of atopic diseases demonstrated a pivotal role of the cytokines interleukin-4 (IL-4) and IL-13, which has spurred the development of tailored therapeutics targeting their common IL-4 receptor (IL-4R). However, several aspects of the IL-4R system remain ill-defined in humans.Whereas all immune cell subsets investigated expressed IL-4Rα and common γ chain protein and mRNA, expression of IL-13Rα1 was restricted to myeloid and B cells. Interestingly, some cells contained considerably more intracellular IL-4R protein than on their surface. Naive B cells were found to carry the highest levels of IL-4Rα distributed evenly between surface and intracellular space, whereas IL-4Rα was found predominantly in intracellular pools in neutrophils. In patients with atopic diseases treated with dupilumab, we observed that engagement of IL-4Rα by dupilumab resulted in internalization of the antibody and decreased total IL-4Rα expression. Notably, even after months of treatment not all intracellular IL-4Rα molecules were occupied by dupilumab, indicating the presence of a “dormant” intracellular IL-4Rα pool that could be mobilized upon certain extrinsic or intrinsic cues.
Collectively, our findings suggest that distinct human immune cell subsets contain surface and intracellular IL-4R pools, which are differently affected by targeted biologic treatment.
Type 2 immune predisposition results in accelerated neutrophil aging causing susceptibility to bacterial infection
Egholm C, Özcan A, Breu D, Boyman O
Science Immunology (2022)
DOI: 10.1126/SciImmunol.ABI9733
Article PDF
Article on ScImm Website
Abstract
Atopic individuals show enhanced type 2 immune cell responses and a susceptibility to infections with certain bacteria and viruses. Although patients with allergic diseases harbor normal counts of circulating neutrophils, these cells exert deficient effector functions. However, the underlying mechanism of this dysregulation of neutrophils remains ill defined. Here, we find that development, aging, and elimination of neutrophils are accelerated in mice with a predisposition to type 2 immunity, which, in turn, causes susceptibility to infection with several bacteria. Neutrophil-mediated immunity to bacterial infection was greatly decreased in mice with a genetic or induced bias to type 2 immunity. Abrogation of interleukin-4 (IL-4) receptor signaling in these animals fully restored their antibacterial defense, which largely depended on Ly6G+ neutrophils.
CCR7-guided neutrophil redirection to skin-draining lymph nodes regulates cutaneous inflammation and infection
Özcan A, Collado-Diaz V, Gunzer M, Halin C, Kolios AGA, Boyman O
Science Immunology (2022)
DOI: 10.1126/sciimmunol.abi9126
Article PDF
Article on ScImm Website
Abstract
Neutrophils are the first nonresident effector immune cells that migrate to a site of infection or inflammation; however, improper control of neutrophil responses can cause considerable tissue damage. Here, we found that neutrophil responses in inflamed or infected skin were regulated by CCR7-dependent migration and phagocytosis of neutrophils in draining lymph nodes (dLNs). In mouse models of Toll-like receptor–induced skin inflammation and cutaneous Staphylococcus aureus infection, neutrophils migrated from the skin to the dLNs via lymphatic vessels in a CCR7-mediated manner.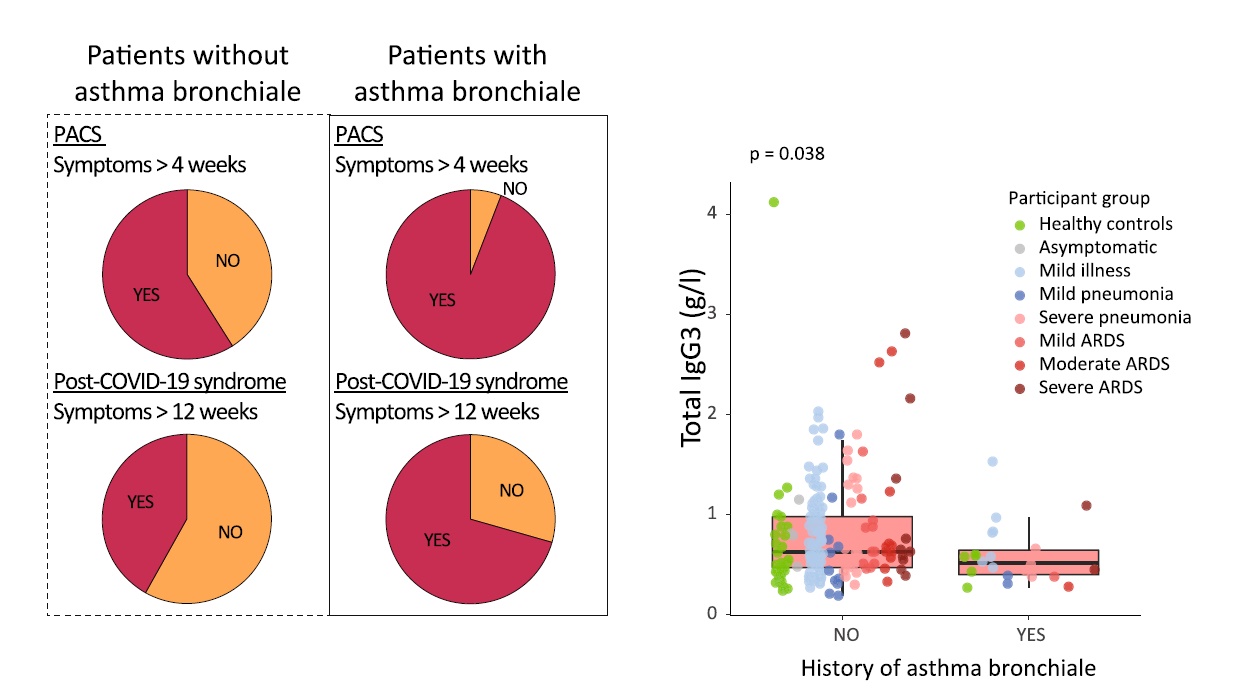
Immunoglobulin signature predicts risk of post-acute COVID-19 syndrome
Cervia C, Zurbuchen Y, Taeschler P, Ballouz T, Menges D, Hasler S, Adamo S, Raeber ME, Bächli E, Rüdiger A, Stüssi-Helbing M, Huber LC, Nilsson J, Held U, Puhan MA, Boyman O
Nature Communications (2022)
DOI: 10.1038/s41467-021-27797-1
Abstract
Following acute infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) a significant proportion of individuals develop prolonged symptoms, a serious condition termed post-acute coronavirus disease 2019 (COVID-19) syndrome (PACS) or long COVID. Predictors of PACS are needed. In a prospective multicentric cohort study of 215 individuals, we study COVID-19 patients during primary infection and up to one year later, compared to healthy subjects. We discover an immunoglobulin (Ig) signature, based on total IgM and IgG3 levels, which – combined with age, history of asthma bronchiale, and five symptoms during primary infection – is able to predict the risk of PACS independently of timepoint of blood sampling.
Signature of long-lived memory CD8+ T cells in acute SARS-CoV-2 infection
Adamo S, Michler J, Zurbuchen Y, Cervia C, Taeschler P, Raeber ME, Baghai Sain S, Nilsson J, Moor AE, Boyman O
Nature (2021)
DOI: 10.1038/s41586-021-04280-x
Abstract
Immunological memory is a hallmark of adaptive immunity and facilitates an accelerated and enhanced immune response upon re-infection with the same pathogen. Since the outbreak of the ongoing coronavirus disease 19 (COVID-19) pandemic, a key question has focused on which severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)-specific T cells stimulated during acute infection give rise to long-lived memory T cells. Using spectral flow cytometry combined with cellular indexing of transcriptomes and T cell receptor (TCR) sequencing we longitudinally characterize individual SARS-CoV-2-specific CD8+ T cells of COVID-19 patients from acute infection to one year into recovery and find a distinct signature identifying long-lived memory CD8+ T cells.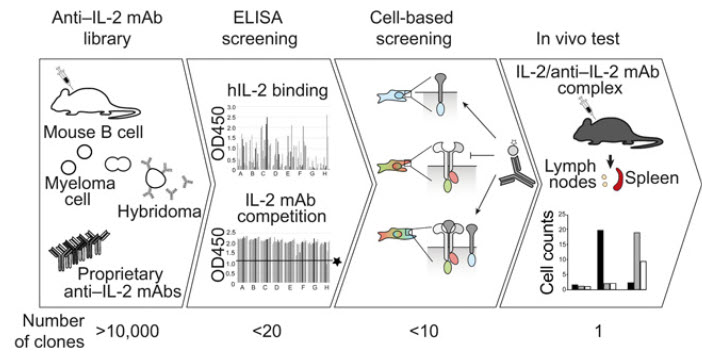
Receptor-gated IL-2 delivery by an anti-human IL-2 antibody activates regulatory T cells in three different species
Karakus U, Sahin D, Mittl PRE, Mooij P, Koopman G, Boyman O.
Science Translational Medicine (2020) 12:eabb9283.
Abstract
Stimulation of regulatory T (Treg) cells holds great promise for the treatment of autoimmune, chronic inflammatory, and certain metabolic diseases. Recent clinical trials with low-dose interleukin-2 (IL-2) to expand Treg cells led to beneficial results in autoimmunity, but IL-2 immunotherapy can activate both Treg cells and pathogenic T cells.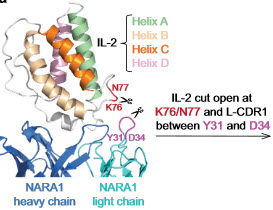
An IL-2-grafted antibody immunotherapy withpotent efficacy against metastatic cancer
Sahin D, Arenas-Ramirez N, Rath M, Karakus U, Hümbelin M, van Gogh M, Borsig L, Boyman O.
Nature Communications (2020) 11:6440.
Abstract
Modified interleukin-2 (IL-2) formulations are being tested in cancer patients. However, IL-2immunotherapy damages IL-2 receptor (IL-2R)-positive endothelial cells and stimulates IL-2Rα(CD25)-expressing lymphocytes that curtail anti-tumor responses.Interleukin-2 signals converge in a lymphoid–dendritic cell pathway that promotes anticancer immunity.
Raeber ME, Rosalia RA, Schmid D, Karakus U, Boyman O.
Science Translational Medicine (2020) 12:eaba5464.
Abstract
Tumor-infiltrating dendritic cells (DCs) correlate with effective anticancer immunity and improved responsiveness to anti–PD-1 checkpoint immunotherapy. However, the drivers of DC expansion and intratumoral accumulation are ill-defined. We found that interleukin-2 (IL-2) stimulated DC formation through innate and adaptive lymphoid cells in mice and humans, and this increase in DCs improved anticancer immunity.Media coverage
This publication has been highlighted in several articles for laypersons:
a. "Botenstoff Interleukin 2 überwindet die Krebsresistenz" ("The cytokine interleukin 2 overcomes cancer resistance"), Schweizerische Depechenagentur and Science APA (2020), 16 September 2020
b. "Forscher umgehen Krebsresistenz" ("Researchers overcome cancer resistance"), 20Minuten (2020), 17 September 2020
c. "Boosting immuno-oncology drugs by recruiting immune messenger cells", FierceBiotech (2020), 21 September 2020
d. "An unconventional cDC outcome of IL-2 immunotherapy", Accelerating Cancer Immunotherapy Research, summary of the main findings and interview of first author Miro Raeber, ACIR webpage 30 September 2020
e. First author Miro Räber explains his main findings in a short research film at JRNL club , April 20, 2021.
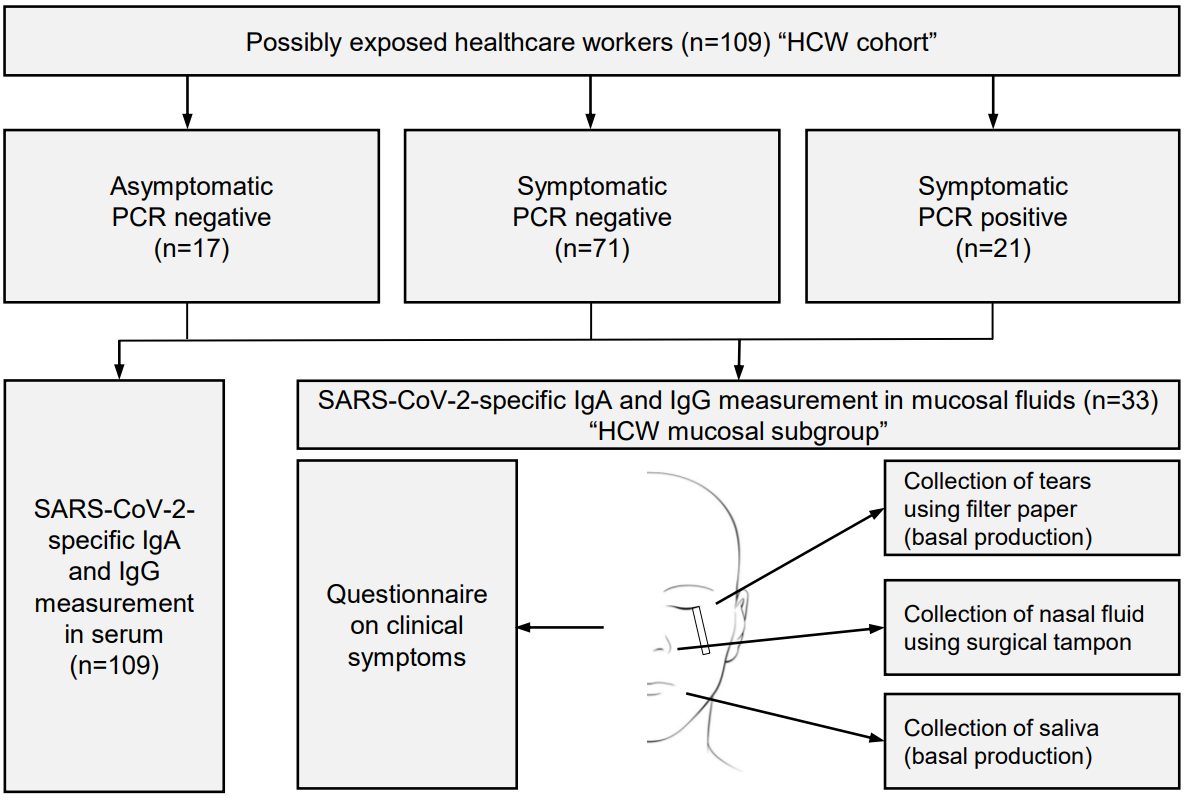
Systemic and mucosal antibody secretion specific to SARS-CoV-2 during mild versus severe COVID-19.
Cervia C, Nilsson J, Zurbuchen Y, Valaperti A, Schreiner J, Wolfensberger A, Raeber ME, Adamo S, Emmenegger M, Hasler S, Bosshard PP, De Cecco E, Bächli E, Rudiger A, Stüssi-Helbling M, Huber LC, Zinkernagel AS, Schaer DJ, Aguzzi A, Held U, Probst-Müller E, Rampini SK, Boyman O.
Journal of Allergy and Clinical Immunology (2020) 147: 545-557.e9.
Abstract
BACKGROUND: Infection with the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes an acute illness termed coronavirus disease 2019 (COVID-19). Humoral immune responses likely play an important role in containing SARS-CoV-2, however, the determinants of SARS-CoV-2-specific antibody responses are unclear.
RESULTS:
INTERPRETATION: These data show that systemic IgA and IgG production against SARS-CoV-2 develops mainly in severe COVID-19, with very high IgA levels seen in patients with severe ARDS, whereas mild disease may be associated with transient serum titers of SARS-CoV-2-specific antibodies but stimulate mucosal SARS-CoV-2-specific IgA secretion. The findings suggest four grades of antibody responses dependent on COVID-19 severity.
Media coverage
This publication has been highlighted in several articles for laypersons:
a. "Wenn der Körper entscheidet, wie schlimm es wird" ("When your body decides how bad it will be"); newspaper article on SARS-CoV-2 antibody tests citing our research article). Der Spiegel (2020), 22 June 2020.
b. "Tests weisen nur einen Fünftel nach" ("Tests might be detecting only a fifth of cases"); newspaper article on SARS-CoV-2 antibody tests based on our research article). TagesAnzeiger (2020), 2 June 2020. Replies in Landbote, Thuner Tagblatt etc.
c. "Nicht nur im Blut hat es Antikörper" ("Antibodies can be found not only in the blood"); newspaper article on SARS-CoV-2 antibody tests based on our research article). NZZ am Sonntag (2020), 31 May 2020.
d. Interview with first author Carlo Cervia on TeleZ (in German, starting 14'04).
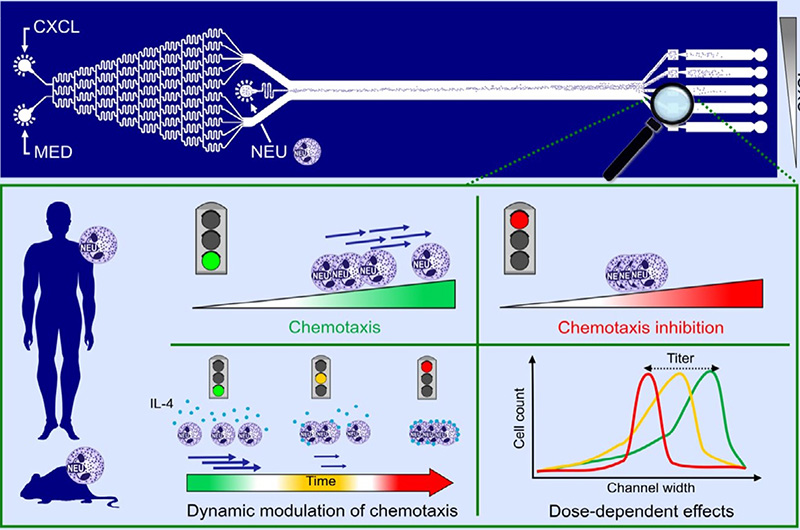
Establishment of a scalable microfluidic assay for characterization of population-based neutrophil chemotaxis.
Grigolato F, Egholm C, Impellizzieri D, Arosio P, Boyman O.
Allergy (2020) 75: 1382-1393.
Abstract
BACKGROUND: Regulation of neutrophil chemotaxis and activation plays crucial roles in immunity, and dysregulated neutrophil responses can lead to pathology as seen in neutrophilic asthma. Neutrophil recruitment is key for initiating immune defense and inflammation, and its modulation is a promising therapeutic target. Microfluidic technology is an attractive tool for characterization of neutrophil migration. Compared to transwell assays, microfluidic approaches could offer several advantages, including precis e control of defined chemokine gradients in space and time, automated quantitative analysis of chemotaxis, and high throughput.
RESULTS:
CONCLUSION: Collectively, our microfluidic device shows several advantages over traditional transwell migration assays and its design is amenable to future integration into multiplexed high‐throughput platforms for screening of molecules that modulate the chemotaxis of different immune cells.
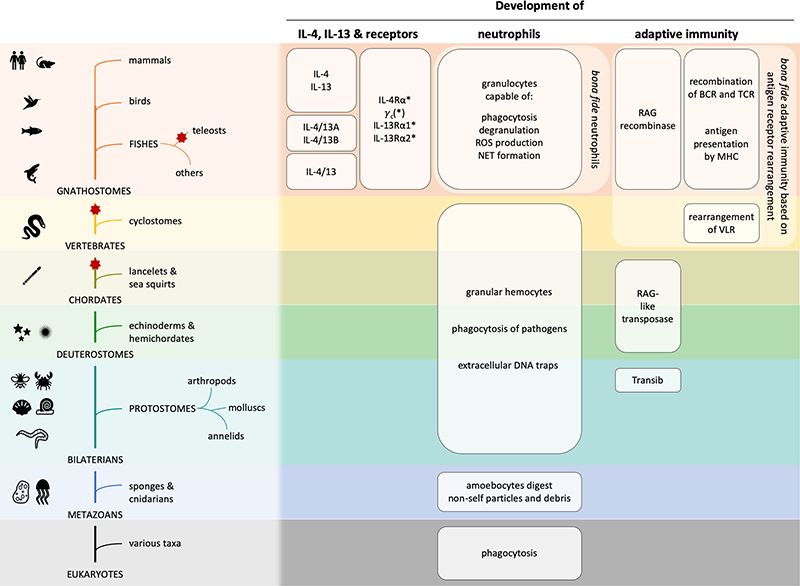
Evolution and function of interleukin-4 receptor signaling in adaptive immunity and neutrophils.
Heeb LEM, Egholm C, Boyman O.
Genes and Immunity (2020) 21:143-149.
Abstract
The cytokines interleukin (IL)-4 and IL-13, signaling via the IL-4 receptor (IL-4R), orchestrate type 2 immunity to helminth infections and toxins. Activation of epithelial and myeloid cells, and a transient neutrophils influx initiates type 2 immune responses, which are dominated by basophils, eosinophils, mast cells, B cell immunoglobulin E production, and type 2 T helper and T follicular helper cells. Interestingly, IL-4 and IL-13 can curtail chemotaxis and several effector functions of neutrophils in mice and humans.
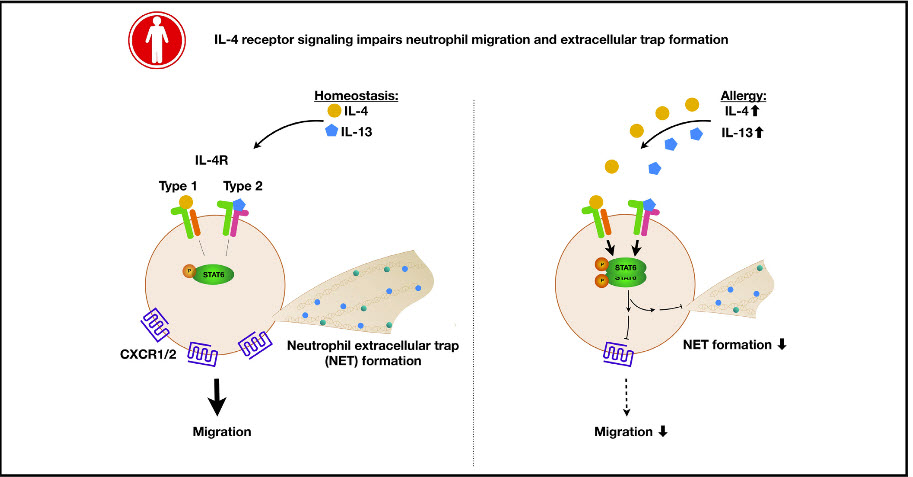
IL-4 receptor engagement in human neutrophils impairs their migration and extracellular trap formation.
Impellizzieri D, Ridder F, Raeber ME, Egholm C, Woytschak J, Kolios AGA, Legler DF, Boyman O.
Journal of Allergy and Clinical Immunology (2019) 144:267-279.
Abstract
BACKGROUND: Type 2 immunity serves to resist parasitic helminths, venoms, and toxins, but the role and regulation of neutrophils during type 2 immune responses are controversial. Helminth models suggested a contribution of neutrophils to type 2 immunity, whereas neutrophils are associated with increased disease severity during type 2 inflammatory disorders, such as asthma.
OBJECTIVE: We sought to evaluate the effect of the prototypic type 2 cytokines IL-4 and IL-13 on human neutrophils.
METHODS: Human neutrophils from peripheral blood were assessed without or with IL-4 or IL-13 for (1) expression of IL-4 receptor subunits, (2) neutrophil extracellular trap (NET) formation, (3) migration toward CXCL8 in vitro and in humanized mice, and (4) CXCR1, CXCR2, and CXCR4 expression, as well as (5) in nonallergic versus allergic subjects.
RESULTS: Human neutrophils expressed both types of IL-4 receptors, and their stimulation through IL-4 or IL-13 diminished their ability to form NETs and migrate toward CXCL8 in vitro. Likewise, in vivo chemotaxis in NOD-scid-Il2rg-/- mice was reduced in IL-4-stimulated human neutrophils compared with control values. These effects were accompanied by downregulation of the CXCL8-binding chemokine receptors CXCR1 and CXCR2 on human neutrophils on IL-4 or IL-13 stimulation in vitro. Ex vivo analysis of neutrophils from allergic patients or exposure of neutrophils from nonallergic subjects to allergic donor serum in vitro impaired their NET formation and migration toward CXCL8, thereby mirroring IL-4/IL-13-stimulated neutrophils.
CONCLUSION: IL-4 receptor signaling in human neutrophils affects several neutrophil effector functions, which bears important implications for immunity in type 2 inflammatory disorders.
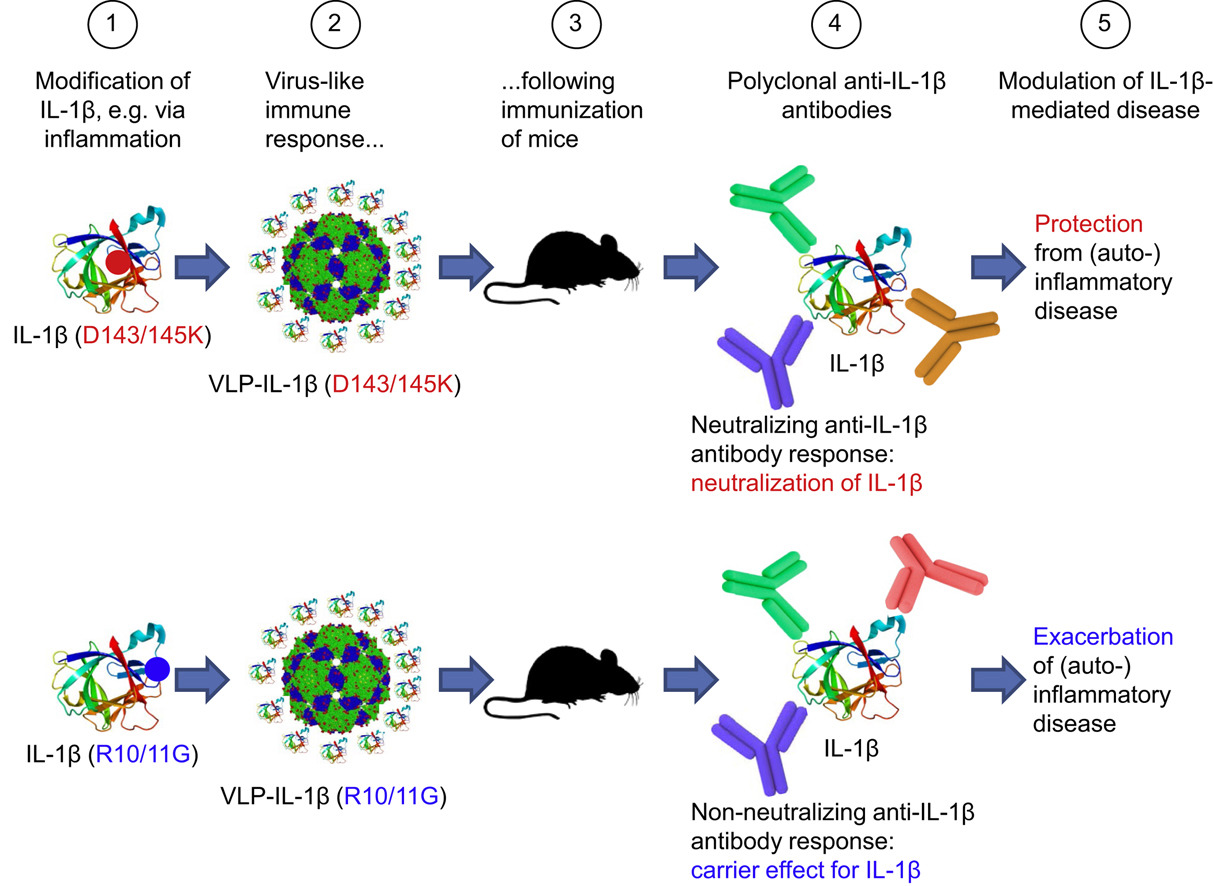
Endogenous polyclonal anti-IL-1 antibody responses potentiate IL-1 activity during pathogenic inflammation
Spohn, G., Arenas-Ramirez, N., Bouchaud, G., Boyman O.
Journal of Allergy and Clinical Immunology (2017) 139:1957-1965.e3
Abstract
Background: Particular neutralizing mAbs to certain cytokines act as agonists in vivo through protection of the cytokine's active site and prolongation of its half-life. Although this principle might be useful for targeted immunotherapy, its role in the pathogenesis of inflammation and autoimmunity is unclear.
Objective: We sought to determine whether slight, structurally nonrelevant modifications of the prototypic proinflammatory cytokine IL-1β during an immune response could elicit polyclonal anti-IL-1β antibody responses that modulated IL-1β's in vivo activity.
Results: Although antibody responses generated to one variant were potently inhibiting IL-1β, antibody responses induced by the other variant even potentiated the in vivo effects of IL-1β; the latter led to enhanced morbidity in 2 different IL-1β-mediated mouse models, including a model of inflammatory bowel disease and an inflammatory arthritis model.
Conclusion: These data demonstrate that endogenous polyclonal anti-cytokine antibody responses can enhance the cytokine's activity in inflammatory and autoimmune diseases.
The Histone Methyltransferase Ezh2 Controls Mechanisms of Adaptive Resistance to Tumor Immunotherapy
Zingg D, Arenas-Ramirez N, Sahin D, Rosalia RA, Antunes AT, Haeusel J, Sommer L, Boyman O.
Cell Reports (2017) 20:854-867.
Abstract
Immunotherapy and particularly immune checkpoint inhibitors have resulted in remarkable clinical responses in patients with immunogenic tumors, although most cancers develop resistance to immunotherapy. The molecular mechanisms of tumor resistance to immunotherapy remain poorly understood.
Improved cancer immunotherapy by a CD25-mimobody conferring selectivity to human interleukin-2
Arenas-Ramirez N, Zou C, Popp S, Zingg D, Brannetti B, Wirth E, Calzascia T, Kovarik J, Sommer L, Zenke G, Woytschak J, Regnier CH, Katopodis A, Boyman O.
Science Translational Medicine (2016) 8:367ra166.
Abstract
Interleukin-2 (IL-2) immunotherapy is an attractive approach in treating advanced cancer. However, by binding to its IL-2 receptor α (CD25) subunit, IL-2 exerts unwanted effects, including stimulation of immunosuppressive regulatory T cells (Tregs) and contribution to vascular leak syndrome.
Type 2 Interleukin-4 Receptor Signaling in Neutrophils Antagonizes Their Expansion and Migration during Infection and Inflammation
Woytschak J, Keller N, Krieg C, Impellizzieri D, Thompson RW, Wynn TA, Zinkernagel AS, Boyman O.
Immunity (2016) 45:172–184.
Abstract
Neutrophils are the first immune cells recruited to sites of inflammation and infection. However, patients with allergic disorders such as atopic dermatitis show a paucity of skin neutrophils and are prone to bacterial skin infections, suggesting that allergic inflammation curtails neutrophil responses.
Full list of publications:
Book Chapters
Interleukin 2
Natalia Arenas-Ramirez, Boyman O.
Encyclopedia of Inflammatory Diseases (Springer). 2015 June 11, pp. 1-9. DOI: 10.1007/978-3-0348-0620-6_132-1
The Role of Interleukin-2 in Memory CD8 Cell Differentiation
Boyman O., Cho JH., Sprent J.
In: Zanetti M., Schoenberger S.P. (eds) Memory T Cells. Advances in Experimental Medicine and Biology, vol 684. Springer, New York, NY DOI: 10.1007/978-1-4419-6451-9_3